Leveraging AI to Work with Cells
Nanofountain Probe Electroporation system enables efficient engineering of stem cells
One of the ultimate goals of medical science is to develop personalized disease diagnostics and therapeutics. With a patient’s genetic information, doctors could tailor treatments to individuals, leading to safer and more effective care.
Recent work from a team of Northwestern Engineering researchers has moved the field closer to realizing this future.
Led by Professor Horacio Espinosa, the research team developed a new version of its Nanofountain Probe Electroporation (NFP-E), a tool used to deliver molecules into single-cells using electricity. The enhanced method leverages artificial intelligence (AI) to execute cell engineering tasks such as cell nuclei localization and probe detection. Other processes such as probe motion, probe-to-cell contact detection, and electroporation-mediated delivery of foreign cargo into single cells are also automated, minimizing user intervention.
“NFP-E can handle small starting samples without any significant cell loss in the entire protocol,” said Espinosa, James N. and Nancy J. Farley Professor in Manufacturing and Entrepreneurship at the McCormick School of Engineering and the study’s corresponding author. “This is an advantage over other cell engineering methods such as bulk electroporation, which require millions of cells and lead to significant cell losses. The automated NFP-E, combined with its ability to selectively target and manipulate single cells in micro-arrays, can be useful in fundamental research, such as deciphering intracellular dynamics and cell-to-cell communication studies as well as biological applications such as cell line generation.”

Espinosa and graduate students Prithvijit Mukherjee, Cesar A. Patino, and Nibir Pathak reported their work in the paper “Deep Learning Assisted Automated Single Cell Electroporation Platform for Effective Genetic Manipulation of Hard-to-Transfect Cells” published March 21 in Small.
“Genetic manipulation of human induced pluripotent stem cells (hiPSCs) by introducing exogenous cargo has a wide range of applications in disease diagnostics, therapeutic discovery, and regenerative medicine,” said Mukherjee, a PhD student in the Espinosa lab who is joining the microfluidics group at Illumina.
Probe-based, microfluidic methods, like NFP-E, use hollow nanopipettes or atomic-force microscopy tips to deliver materials into cells. NFP-E also allows researchers to selectively manipulate cells of interest, work with very small starting samples, and deliver both proteins and plasmids in a variety of animal and human cell types with dosage control.
“The challenge with probe methods, however, is that they require manual operation and produce low throughputs, making them unsuitable for common cell engineering workflows,” said Patino, a PhD student in the Espinosa group.
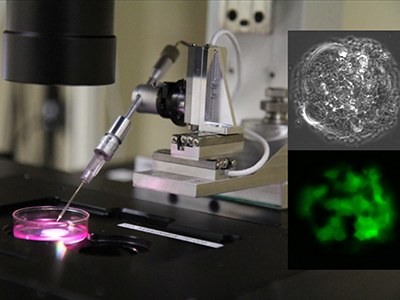
“Selective cell manipulation at sufficient throughput is challenging,” Espinosa said. “Most methods either provide high throughput at the expense of individual cell control or sacrifice throughput for single cell selectivity and control.”
This new work changes that.
The research team’s automated NFP-E enables selective cell engineering at higher-throughputs than manual probe-based methods while also reducing experimental variability and enabling more efficient engineering of hiPSCs. Using the automated platform NFP-E, Espinosa and his colleagues delivered clustered regularly interspaced short palindromic repeats (CRISPR) RNP to hiPSCs for efficient knockout of genes in a variety of culture formats: culture plates, micro-patterns, and micro-wells arrays. The automated engineering of cells in micro-arrays using the NFP-E has potential applications such as isogenic cell line generation from single cells and studying dynamic cellular processes such as intracellular signaling cascades and cell-cell communication.
Espinosa and his team will next work to automate NFP-E’s entire workflow, which includes steps such as automated cell imaging, cell tracking, switching probes, and media exchange for cell culture.
“The idea is to establish a fully automated cell line generation workflow using the combination of the NFP-E and the micro-well arrays,” Espinosa said. “The AI can be further trained to recognize and target specific cell types in multi-cell co-cultures. This can be useful in understanding dynamics such as disease progression or cell communication.”
About this study
The research was supported by two National Institutes of Health grants, awards number 1R43GM128500-01 and 1R21GM132709-01.