In the Fight Against Cancer, a Closer Look at Nuclear Blebbing
Researchers gain insight into abnormally shaped cell nuclei of people with cancer and other disorders
Misshapen cell nuclei are frequently observed in the cells of people with cancer and other diseases, but what causes the abnormality — and why it is associated with certain disorders — has remained unclear.
Researchers at Northwestern University have recently developed a mathematical model that sheds light on the defect by clarifying the mechanisms that cause bulges known as “blebs” in cells’ nuclear membranes. The research — a collaboration between experts at the McCormick School of Engineering and Applied Science and the Feinberg School of Medicine — could be a step toward bleb prevention, which may ultimately provide potential therapies for related diseases.
A paper describing the research, titled “Mechanical Model of Blebbing in Nuclear Lamin Meshworks,” was published Feb. 11 in the Proceedings of the National Academy of Sciences USA (PNAS).
“Changes in the shape of the nucleus are indicative of a range of pathologies, including the premature aging disorder Hutchinson-Gilford progeria syndrome, Emery–Dreifuss muscular dystrophy and some cancers,” said Monica Olvera de la Cruz, the Lawyer Taylor Professor of Materials Science and Engineering, Chemistry, and (by courtesy) Chemical and Biological Engineering at the McCormick School and the corresponding author of the paper. “Our research suggests that blebbing may be the result of an imbalance between the various proteins that constitute the nuclear lamina.”
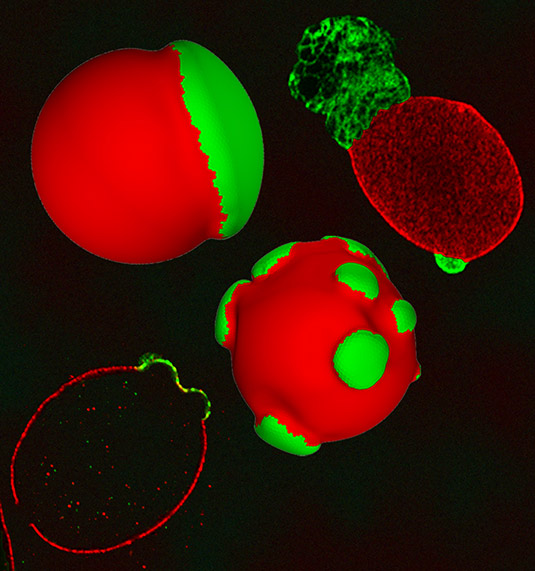
The nucleus — the control center of the cell, the keeper of genetic material and overseer of cell growth and reproduction — is covered by a nuclear envelope consisting of a double membrane and an underlying structure called the nuclear lamina that surrounds the surface of the nucleus and gives it shape. In addition to its mechanical support, the lamina helps regulate cell division and organize genetic material.
In the majority of healthy cells, the nucleus appears smooth and maintains an overall spherical shape, but abnormal nuclear shapes characterized by blebs have been observed in the cells of people suffering from some forms of cancer and other diseases.
In mammals, the lamin meshworks that make up the nuclear lamina consist of mainly two types of lamin proteins, known as types A and B, which are wrapped like two nets around the nucleus. Under normal conditions, the A-type and B-type lamins co-exist throughout the sphere, creating a healthy lamina of approximately even thickness throughout.

But when one of the B-type lamins is depleted, researchers found the A-type and B-type lamins begin to segregate from one another, resulting in an uneven mesh layer with altered mechanical properties. In some regions, the lamina’s fibers begin to gap and separate, giving rise to nuclear blebs, bulges in the cell’s nuclear envelope.
The nuclear lamins, especially the A-type lamins, are now considered to be major building blocks of nuclear architecture and are thus involved in numerous important nuclear functions. Much of the recent information on the functions of the nuclear lamins comes from findings demonstrating that many different human diseases are caused by hundreds of mutations in the nuclear lamin A gene. Many of these diseases are accompanied by changes in nuclear shape and altered lamin organization.
“This study helps us to begin to understand how these abnormal shapes are formed,” said Robert D. Goldman, the Stephen Walter Ranson Professor of Cell and Molecular Biology, chair of the department of cell and molecular biology at the Feinberg School, and one of the paper’s authors. “Collaborations between physicists and cell biologists are beginning to reveal new insights into these normal and abnormal cells.”
Enabling some of those new insights, the Northwestern researchers designed an energy-minimizing continuum elastic model that enabled them to produce structures with comparable shapes and patterns as those found in naturally occurring pathological nuclei.
Other co-authors of the paper are Chloe M. Funkhouser (lead author) and Rastko Sknepnek from McCormick’s department of materials science and engineering and Takeshi Shimi and Anne E. Goldman from Feinberg’s department of cell and molecular biology.